
by Health and Environmental Science Editor Dr. Ahimsa Porter Sumchai
It is a rare moment indeed at the Hunters Point Shipyard when the U.S. Navy publicly admits it has uncovered a dangerous bone-seeking radionuclide with a half-life of 28.9 years in concentrations exceeding those set to protect human life and safety at a federal Superfund site.
Strontium-90 is formed by nuclear explosions and is considered to be the most dangerous component of nuclear fallout by the federal Agency for Toxic Substances and Disease Registry. High levels of radioactive strontium damage bone marrow and cause anemia and blood clotting disorders.
Navy chemists detected Strontium-90 in samples obtained on a shipyard parcel in concentrations above the remediation goal set for public safety. When they reanalyzed two of the samples – voila! – the high Strontium-90 concentrations magically went away!

That would be the end of the story except that guided by EPA and regulatory input, the Navy has decided to “refine” its methods of Strontium-90 detection by increasing the size of the soil sample to 2.5 grams and extend the laboratory analysis by seven days. The Navy will reanalyze all samples using this updated method according to a fact sheet released to the public on Oct. 21, 2021.
The Department of the Navy is a military organization – not a public health organization. The mission of the Navy is to “maintain, train and equip combat-ready naval forces capable of winning wars, deterring aggression and maintaining freedom of the seas.”
There is no safe level of radiation.
The Navy fact sheet concludes by asking if you should be concerned about Strontium-90 at the shipyard: “No. Strontium-90 lab results to date have not indicated levels considered a risk to human health or the environment.”
Now that is one helluva oxymoronic statement! There is no safe level of radiation. That is the simple conclusion of the Committee on the Biological Effects of Ionizing Radiation in its research on the effects on populations of exposure to low levels of ionizing radiation, health effects and health risks from exposure to low levels of ionizing radiation from 1980 to 2006 https://www.euronuclear.org/glossary/beir/.
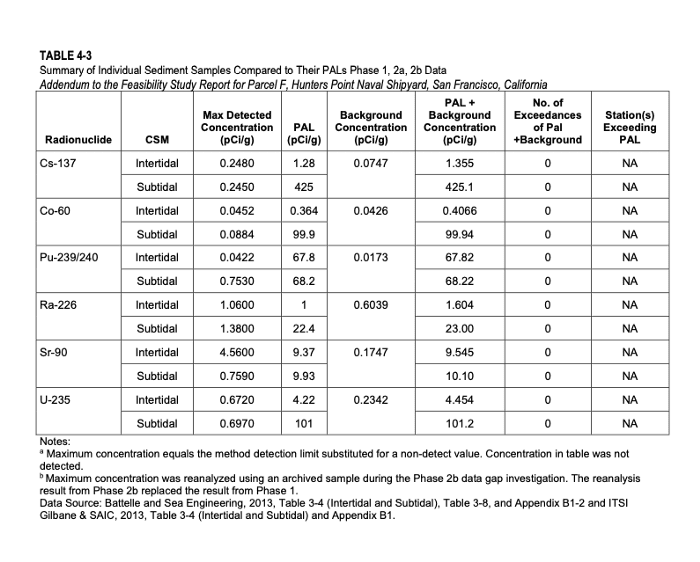
Finding Strontium-90 in concentrations exceeding remediation goals in 10 percent of samples is conclusive evidence of risk to human health. If this were not the case, the Navy would not be reanalyzing all of the samples collected in the region of concern.
“The US National Academy of Sciences published [in 2006] the Biological Effects of Ionizing Radiation (BEIR) VII report, which concluded that the existing scientific evidence is consistent with [LNT, the linear no-threshold model of radiation-induced cancers].” “According to the LNT, every fraction of ionizing radiation, no matter how small, constitutes an increased cancer risk – linear with the dose. This model is the basis for current radiation regulation.” This via the article “Health Impacts of Low Dose Ionizing Radiation: Current Scientific Debate and Regulatory Issues.”

Strontium-90 is a bone-seeker. It is chemically similar to calcium and when inhaled your body deposits it in your bones, where it emits damaging beta radiation particles to your bone marrow. Beta radiation is emitted within a short range, making it most harmful if inhaled or swallowed, and it may lead to bone cancer or leukemia. A toxicological profile for Strontium-90 was developed by ATSDR that examined biomarkers of exposure and effect and methods for reducing toxic effects.
Strontium-85 emits gamma rays. It has been abandoned from use to detect bone uptake in radiographic imaging. Radioactive strontium can be absorbed into the bones of small children. Large doses replace calcium in bone and lead to renal failure, bone deformity and tumors.
According to “EPA Facts About Strontium-90,” the most common isotope of strontium is Strontium-90. Radioactive Strontium-90 is produced when uranium and plutonium undergo fission. Fission is the process whereby the nucleus of a radionuclide breaks down into smaller parts. Large amounts of radioactive strontium were produced during nuclear weapons testing.
The half-life is the time required for a radioactive substance to lose 50 percent of its radioactivity by decay. Strontium is not stable, and the release of radiation caused by its decay is a concern because beta particles can pass through skin.
Watch Video: https://youtube.com/shorts/3T-5cOptm54?feature=share
Of greater concern is the ability of strontium to become part of the food chain particularly in calcium containing dairy products like milk and cheese. The EPA has made recommendations to protect human health at Superfund sites in the fact sheet “Primer on Radionuclides Commonly Found at Superfund Sites.” The EPA has established a Maximum Contaminant Level (MCL) of 4 millirems per year for beta particles from man-made radionuclides.
SF Bay View Health and Environmental Science Editor Ahimsa Porter Sumchai, MD, PD, founder and principal investigator for the Hunters Point Community Biomonitoring Program, founding chair of the Hunters Point Naval Shipyard Restoration Advisory Board’s Radiological Subcommittee and contributor to the 2005 Draft Historical Radiological Assessment, can be reached at AhimsaPorterSumchaiMD@Comcast.net. Dr. Sumchai is medical director of Golden State MD Health & Wellness, a UCSF and Stanford trained author and researcher, and a member of the UCSF Medical Alumni Association Board of Directors.







